
Thomas Jefferson University Hospital Division of Neuroradiology
Sunday, March 29, 2026
Research Environment, TJUH Neuroradiology


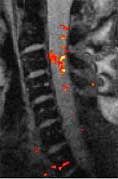
The team has a number of clinical and basic research projects underway, including advanced imaging of cerebral ischemia, spinal cord injury, Alzheimer's Disease, brain segmentation, MR parameter estimation, flow analyisis, cerebral neoplasia, gene therapy of human glioma, DWI/PWI, magnetization transfer, Multi-voxel proton MR spectroscopy in temporal lobe epilepsy, functional neuroimaging, expert systems, and outcomes analysis. We are also one of the first sites to reliably measure the fMRI signal from human spinal cord using a commercial 1.5T magnet.
Research In the Jefferson MRI Physics Laboratory
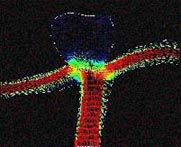
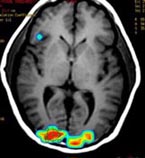
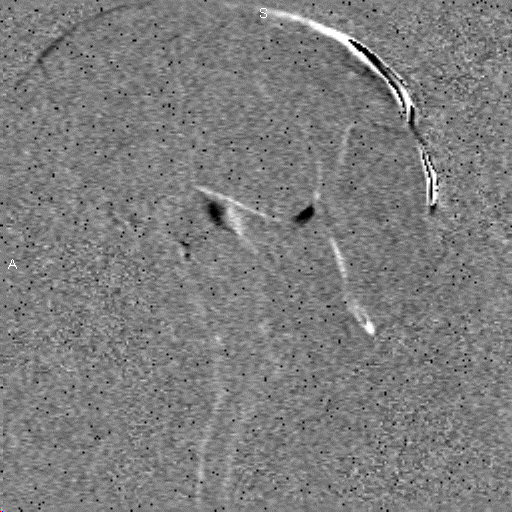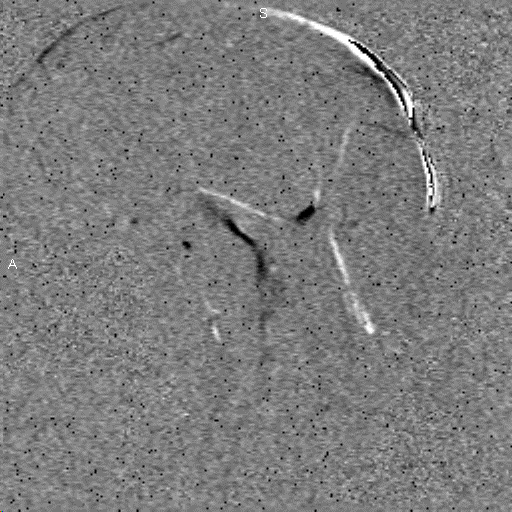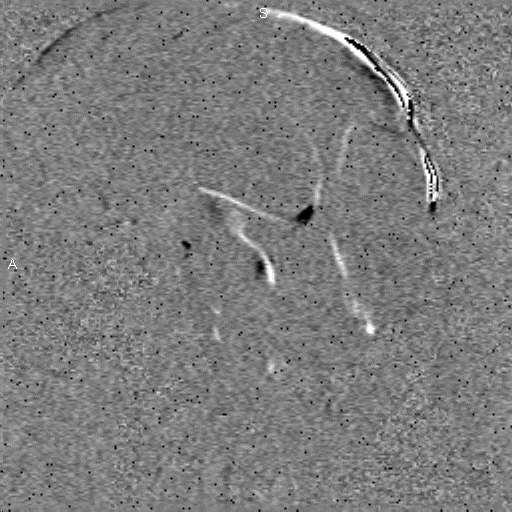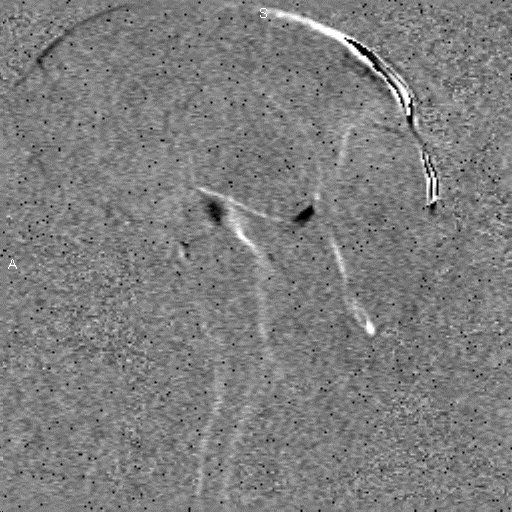
Research in the MRI Physics Laboratory is devoted to the advancement of magnetic resonance imaging (MRI) for applications in neurofunctional imaging and diagnostic radiology, with focus being on brain functional MRI (fMRI). Going beyond the traditional strength of MRI for structural imaging, fMRI allows for imaging brain at activity by detecting cerebral hemodynamic responses (i.e., perfusion and oxygenation) accompanying brain activation. Research in the Lab involves combining analytical and experimental methods of MRI physics and engineering with biomedical sciences to achieve a better understanding of human brain function. Specifically, brain function in health and disease is investigated through the following angles:
Quantitative characterization of cerebral hemodynamics, including changes in perfusion and oxygenation, accompanying brain activation or due to a disease. This involves development of robust MRI pulse sequences for efficient and accurate measurement of these physiological indices, as well as modeling calculation for improved interpretability of fMRI signals. Of particular interest is the development of pulse sequences that allow for simultaneous acquisition of both BOLD (blood oxygenation level dependent) and perfusion images, which in turn allows for assessment of relative change (either due to brain activity or a disease) in cerebral metabolic rate in oxygen (CMRO2), a more direct index of brain function. One unique strength of these techniques is their ability for acquisition/display of cerebral blood flow (CBF) maps without the need of offline post-processing, which can find clinical applications such as in acute ischemic stroke MRI, because the physicians can view the CBF maps on screen immediately after the scans are done.
Study of brain functional architecture. This involves combining delineation of brain structural connectivity via diffusion tensor imaging (DTI) with investigation of brain functional connectivity via studying low-frequency spontaneous oscillation of regional blood flow and oxygenation. It has been found that there exists strong correlation between low-frequency spontaneous oscillation of resting state BOLD signals of spatially separated but functionally connected brain areas, which makes it possible to construct brain functional connectivity maps. DTI is a new, fast evolving technique for imaging and assessing tissue water diffusivity, diffusion anisotropy, and white matter fiber tract integrity. Addition of a magnetic field gradient makes NMR signal sensitive to water molecule diffusion arising from thermal motion or Brownian motion along a direction of interest. Brain microstructure forms physical boundaries that constrain water diffusion making it direction-dependent, i.e., anisotropic. White matter contains organized fiber bundles making water diffuse along the axis of WM pathways. We are interested in mapping axonal connections between cortical regions and cortical disconnection caused by brain lesions, epilepsy, aging and aging-related diseases, and substance abuse by combining functional connectivity maps with diffusion tensor fiber tractography.Study of the vascular imprints of brain activity. SWI develops image contrast based on the bulk magnetic susceptibility difference between tissues. Application of our high spatiotemporal resolution SWI sequence in brain tumor patients have revealed exquisite details of the tumor venous vasculature that are not visible with other techniques. In addition to application for imaging venous vasculature, SWI has also been demonstrated to be a powerful tool for differentiating tissues with small differences in iron content, for water/fat separation, and for visualizing clot and microhemorrhage. We are interested in developing high spatiotemporal resolution SWI to map the vascular imprints of brain activity.
The above techniques have been explored in patient studies. For example, we are currently carrying out a project in which DTI and functional connectivity are combined to assess cerebral neoplasia in clinical brain tumor patients. Three distinct patterns have been observed: 1) when white matter fibers were not apparently affected by a lesion, functional connectivity was largely maintained; 2) when a lesion caused displacement of white matter tracts without significantly affecting diffusion anisotropy, functional connectivity was found to be correspondingly distorted; 3) when white matter tracts were disrupted, functional disappeared too. These results provided evidence that DTI represents a robust tool to non-invasively examine in vivo structural connectivity underlying functional connectivity. In collaboration with cancer researchers, the Lab has also applied quantitative MRI protocols in human cancer xenograft models in athymic nude mice. Preliminary results suggest that MRI transverse relaxometry and tissue diffusivity mapping represents a novel non-invasive imaging modality for delineating in vivo tumor pathophysiological characteristics related to oxygen metablism, angiogenesis, and growth, all of which are important clinical diagnostic parameters.
Copyright © 2019 Thomas Jefferson University. All Rights Reserved.
The Thomas Jefferson University web site, its contents and programs, is provided for informational and educational purposes only and is not intended as medical advice nor is it intended to create any physician-patient relationship. Please remember that this information should not substitute for a visit or a consultation with a health care provider. The views or opinions expressed in the resources provided do not necessarily reflect those of Thomas Jefferson University, Thomas Jefferson University Hospital, or the Jefferson Health System or staff.